Developing safer, more effective and more usable medical products hinges upon robust human factors engineering (HFE) processes and expertise. HFE encompasses substantial efforts such as user research and analysis, design work and product evaluation, but health and life sciences companies should not overlook the crucial role instructions for use (IFU) can play in supporting product safety and usability.
We talk with Michael Wiklund, principal consultant, Human Factors Research & Design at Emergo by UL, about the benefits and challenges of developing effective IFUs that can help improve interactions of patients and users with medical products and technologies.
Before we go into the specifics, can you share some trends or shifts you foresee in Medtech product development in 2025 and the next few years?
Despite its reputation for slow movement, the medical device industry seems to be in a particular dynamic phase, at least from a user interface design perspective. Many devices that used to function in a standalone manner are now part of connected systems or components. Such systems may have autonomous and AI-enabled capabilities that keep patients in touch with medical care providers.
As a result, user interfaces have become a bit more complicated and often require users to have a modicum of internet access and surfing capability. Also, many patient diagnostic, therapeutic and monitoring solutions that lived in clinical environments have migrated into people’s homes. This represents progress in terms of patient convenience but puts added pressure on manufacturers to make their medical products more intuitive to use. This is where good instructions for use can be a vital complement to a good user interface.
With the evolving trends in Medtech development, how does an IFU fit into the larger scheme of applying human factors during the medical product development process?
Well-written and smartly designed instructions for use (IFUs) are absolutely worth the development effort. An excellent IFU can contribute to the safe, effective and satisfying use of a product. Conversely, a poor IFU can frustrate users and possibly induce harmful use errors.
Producing an excellent IFU starts with determining users’ needs and preferences regarding the job aid. Then, developers need to follow best practices in terms of writing and document design. Finally, preliminary designs should be tested, nominally by conducting a usability test involving the intended users who engage in use scenarios involving the IFU. An excellent IFU can help a product meet a given industry’s standard of care, satisfy human factors regulations that may apply and drive a product’s commercial success by generating enhanced customer goodwill.
What has been the common barrier to developing an effective IFU?
IFUs are an oft maligned part of a product’s user interface, which involves a set of human touchpoints that may also include hardware, software, labels and packages. Why maligned? It’s because people often prefer to rely upon experience, common sense, trial and error and tips from a friend to use an unfamiliar product or perform an unfamiliar task with a product they have been using for a while. They are less inclined to read a product’s accompanying instructions, that is if any are available when needed, to guide them.
This inclination has led many product developers to conclude that people do not read instructions. In turn, this leads developers to limit their investment when it comes to producing IFUs. The predictable results, arising from the negative reinforcement loop, have been impoverished IFUs that are neither particularly well-written nor smartly designed. This is a shame for the product manufacturer and consumers because IFUs can be a vital asset to both parties.
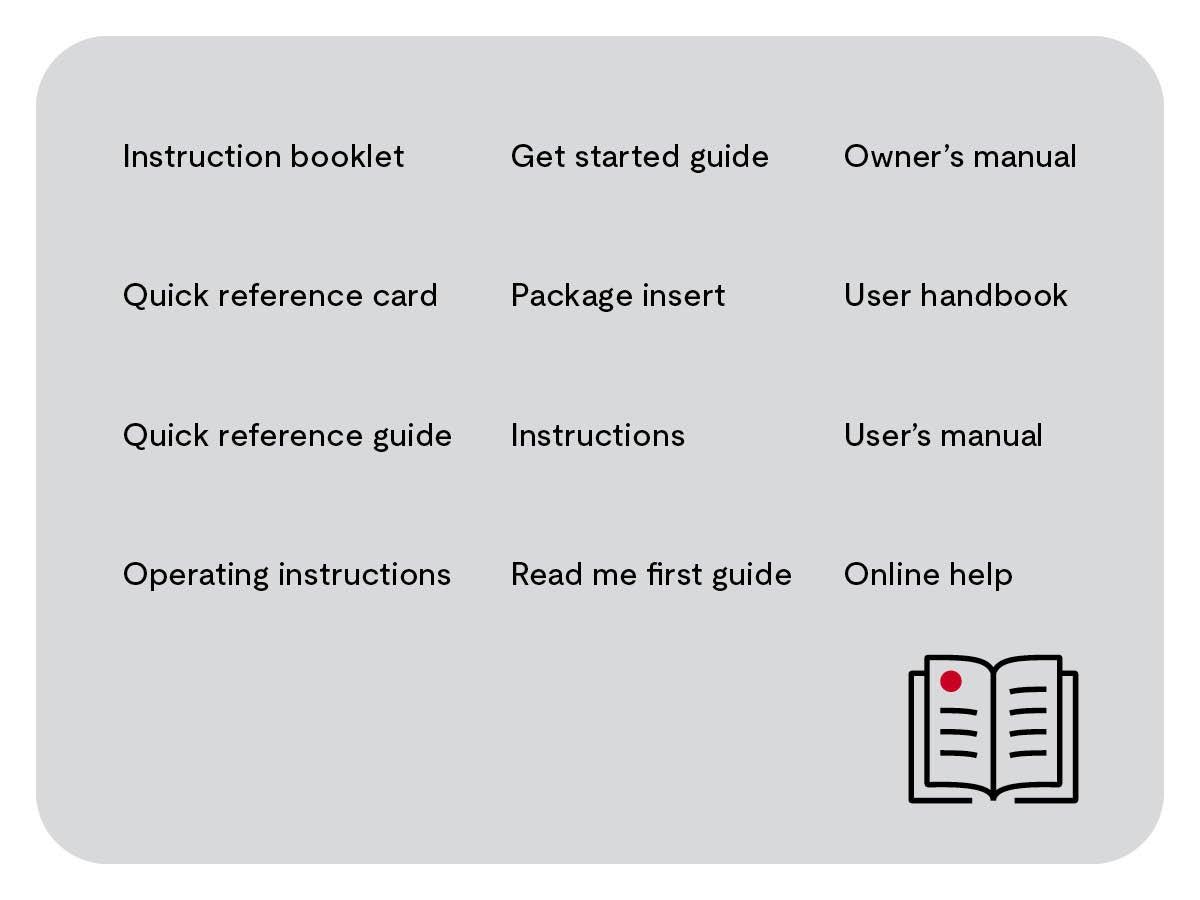
IFUs go by many names (shown below). The names tell you something about the product, the instructional goal, the amount of content and the communication medium (e.g., printed, online).
How does an effective IFU contribute to product safety, effectiveness and/or usability?
A manufacturer that strives to produce a good IFU can help people in two ways: (1) get the best performance out of their product, and (2) help people avoid making mistakes that might not only be annoying but also harmful. As such, a good IFU can create added value for the customer, translating into goodwill and positive reviews of the product. It also has the potential to reduce product complaints and returns, as well as reduce a manufacturer’s vulnerability to product liability claims.
Sure, some people will skip reading an IFU in favor of “giving it a go” with a new product, particularly simple ones that are not likely to cause harm or be broken and expensive to fix. However, when products are more complicated, potentially dangerous if operated improperly and expensive to repair, many consumers will consult an IFU. At this point, and if the IFU is a good one, users should be rewarded by information putting them on the right course of action. A poorly written and designed IFU is less likely to be helpful and will reinforce the stereotype that “instructions are not worth the paper on which they are printed.”
How can medical product developers improve users’ receptivity to IFUs?
In usability tests of IFUs, most participants — perhaps two-thirds — resort to reading an IFU only when they encounter a problem or are uncertain about the next step. This is not when people are feeling relaxed and patient. It’s quite the opposite scenario. And so, an IFU has a brief window of time to demonstrate value. A well-designed IFU does this quickly. A poorly designed IFU will cause someone to emit a discouraged sigh or worse and set the IFU aside for good.
As more product developers embrace IFUs as a valuable resource, society is likely to see better ones, which will raise user expectations. The negative reinforcement loop will become a positive one.
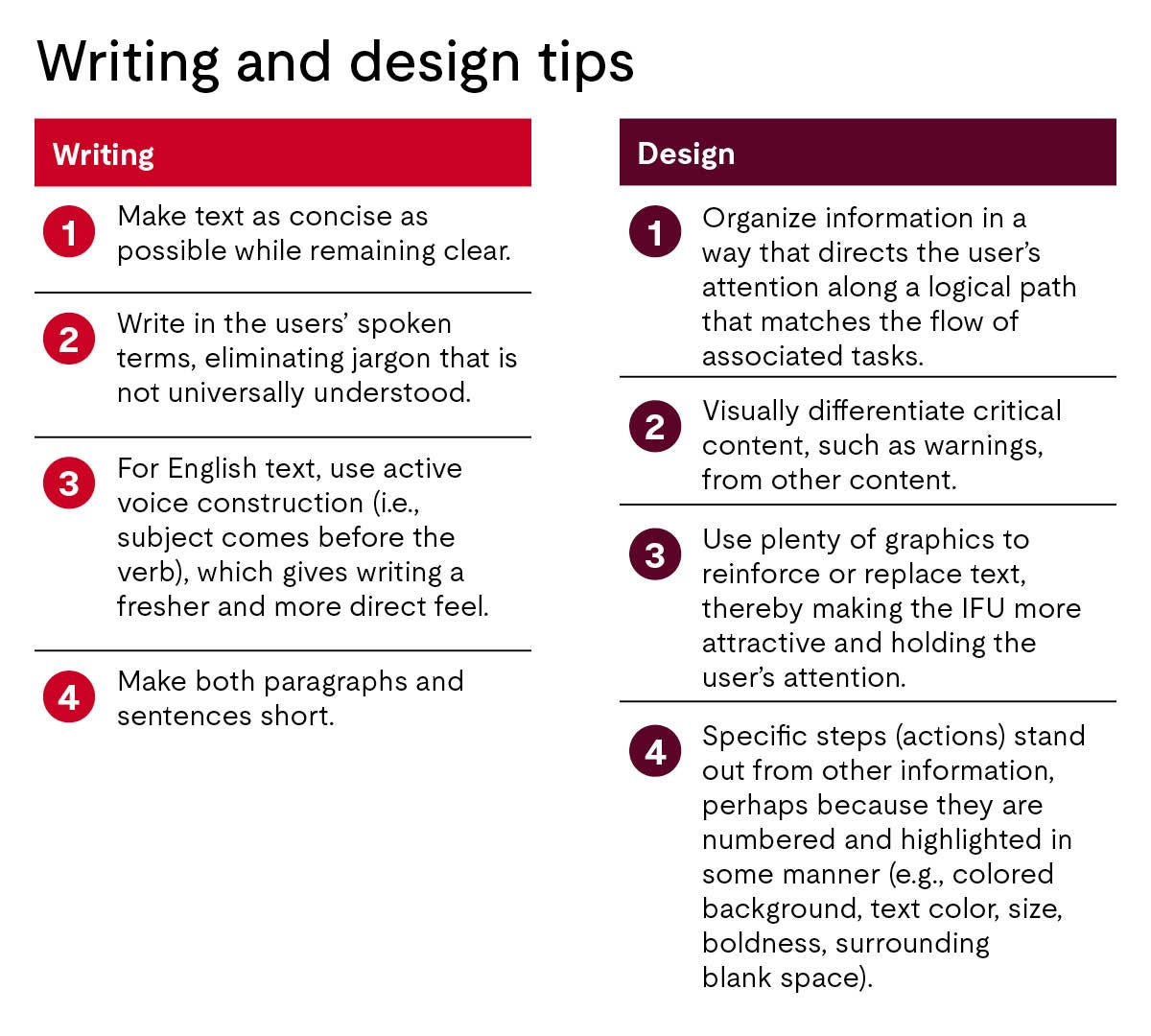
Any closing thoughts on making IFUs more effective?
When a manufacturer invests considerable effort into producing an excellent IFU, it shows respect for the product users. The manufacturer wants the consumer to have a great user experience enhanced by well-written and well-designed instructions. That is why an excellent IFU generates goodwill toward a product developer. It can have a “halo effect.”
It’s true that products, such as today’s smartphones, don’t come with instruction booklets, and that some consumers are impressed by this. But keep in mind that the same people might use their laptop computer to find online instructions on how to do something on their smartphone. Meanwhile, products as diverse as automobiles, dishwashers, power tools and blood pressure monitors come with instructions — and for good reason.
Within UL Solutions we provide a broad portfolio of offerings to all the medical device industries. This includes certification, Approved/Notified Body and consultancy services. In order to protect and prevent any conflict of interest, perception of conflict of interest and protection of both our brand and our customers brand, we have processes in place to identify and manage any potential conflicts of interest and maintain impartiality.
Become industry innovators of safe, compliant, sustainable products
Our safety science expertise and software enable industry innovators across the medical product lifecycle to overcome critical challenges and develop safer, more effective products that empower users globally.
Get connected with our sales team
Thanks for your interest in our products and services. Let's collect some information so we can connect you with the right person.